Projects
01
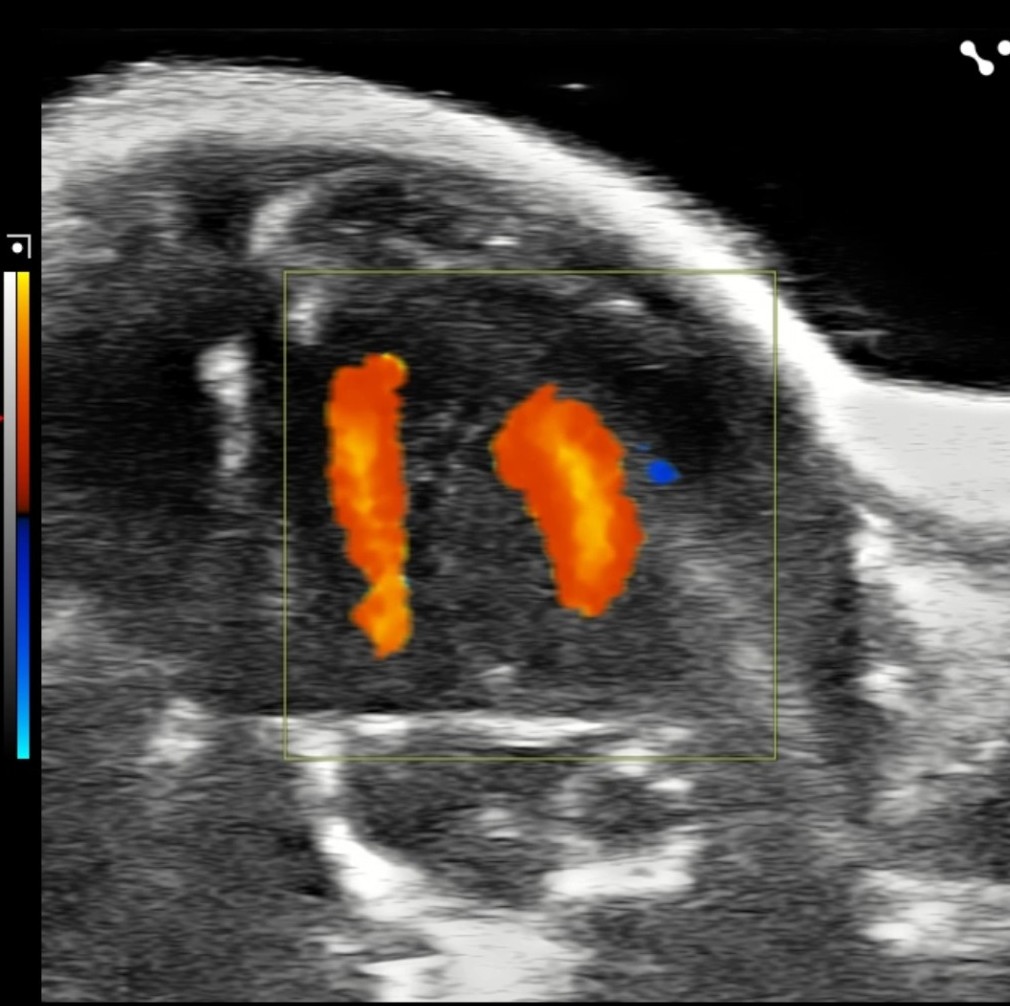
Targeted Cardiac Drug Delivery
My research focuses on developing targeted cardiac drug-delivery strategies using the intrapericardial space as a novel and clinically relevant therapeutic access route. I established an intrapericardial catheter–based platform that enables direct, localized delivery of small molecules, biologics, nanoparticles, and cell-based therapies to the heart, allowing precise modulation of cardiac biology while minimizing systemic exposure.
This approach has gained rapid visibility following its initial publication, prompting multiple independent commentaries and underscoring strong interest from both clinical and basic research communities. Its early adoption highlights the platform’s novelty, translational potential, and relevance to cardiovascular therapeutics.
Building on this foundation, my work aims to expand this delivery system into a versatile preclinical pipeline for evaluating next-generation therapies targeting immune regulation, angiogenesis, fibrotic remodeling, ischemia/reperfusion injury, and pericardial inflammation in cardiovascular disease.
02
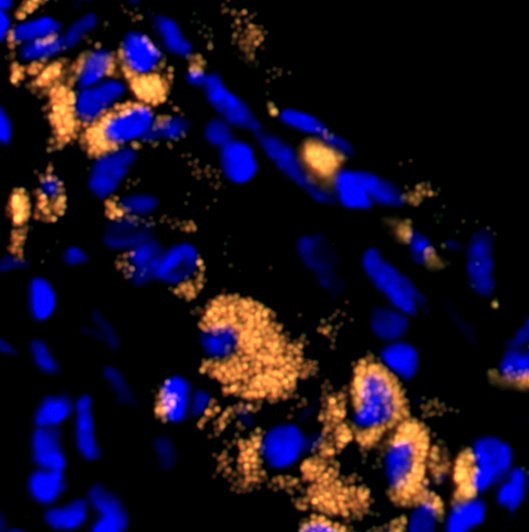
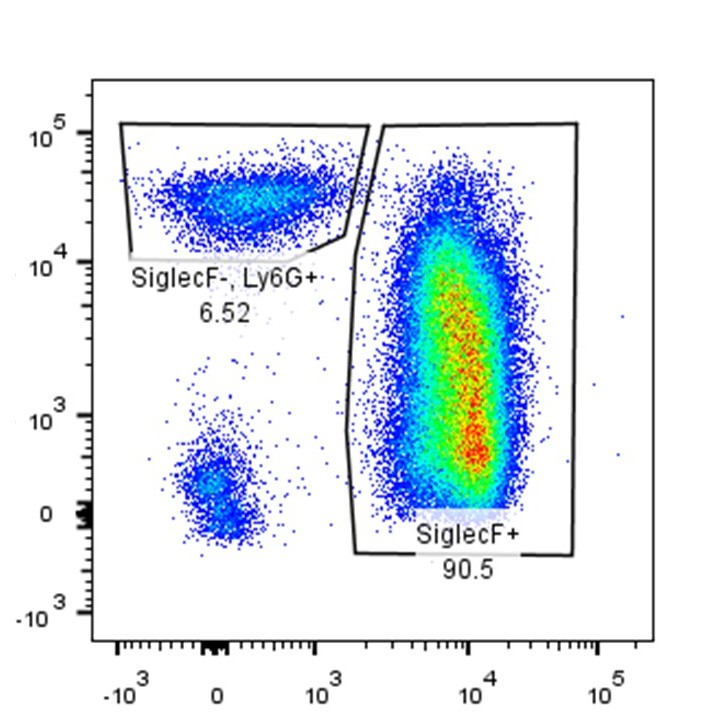
Immune Mechanisms in Cardiac Injury and Repair
A central pillar of my research program is the investigation of immune-mediated mechanisms underlying myocardial ischemia–reperfusion (I/R) injury in related cardiac pathologies. I have modified and optimized murine models of myocardial ischemia–reperfusion to enable unbiased, reproducible studies of immune responses following cardiac ischemic injury, allowing precise temporal dissection of inflammatory and reparative phases after prolonged ischemia–reperfusion.
Using this platform, I have defined the molecular and cellular dynamics of innate and adaptive immune responses following myocardial ischemia–reperfusion and characterized how immunomodulatory interventions, including cyclosporine A, influence immune activation and functional outcomes after myocardial infarction.
In parallel, my work identified cardiac myosin–specific tissue-resident memory T cells as critical drivers of immune checkpoint inhibitor–associated myocarditis using a clinically relevant two-hit mouse model of immune reactivation, providing mechanistic insight into how maladaptive immune memory triggers severe cardiac inflammation.
Together, these studies establish a unified framework for understanding immune-driven cardiac injury and repair and support the development of targeted strategies to improve outcomes of heart disease.
03
Cardiac Valve Pathobiology
A third major pillar of my research focuses on the mechanisms that drive cardiac valve remodeling and degenerative valve disease. My work has demonstrated that mechanosensitive signaling through the ion channel PIEZO1 alters serotonin receptor pathways and promotes collagen synthesis in human mitral valve interstitial cells, linking biomechanical forces to fibrotic valve remodeling.
To extend these findings in vivo, I developed a murine model of serotonin transporter–dependent mitral valvopathy and established key experimental approaches for quantitative assessment of mitral valve structure and function in mouse models. Together, these platforms enable integrated investigation of cellular signaling, tissue remodeling, and disease progression in the mitral valve.
Collectively, these studies provide a mechanistic and translational framework for understanding degenerative mitral valve disease and for designing targeted interventions aimed at modulating mechanosensing, serotonin signaling, and fibrotic remodeling.
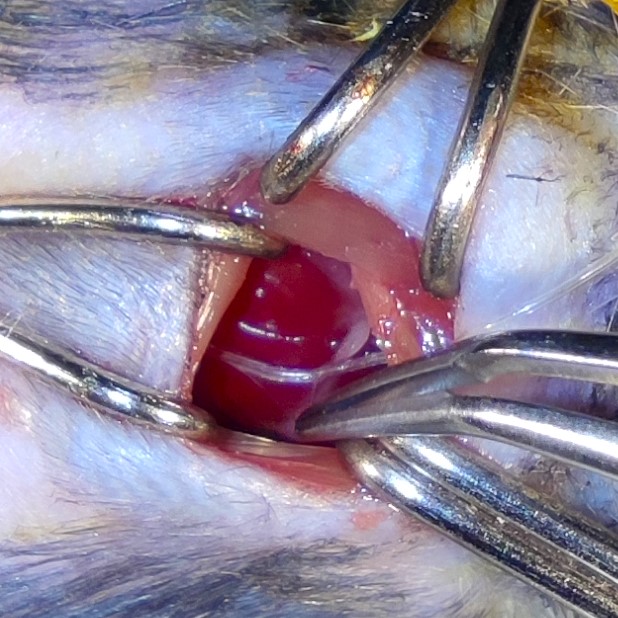
04
Advanced Preclinical Modeling for Cardiovascular Diseases
One of the central focuses of my research program is the development and refinement of minimally invasive, mechanistically informative murine models of cardiovascular disease. Many widely used small-animal models remain biased or insufficiently representative of human pathology, limiting their translational value and interpretability. My work addresses these limitations through systematic optimization of models for ischemia–reperfusion injury, post-infarction remodeling, degenerative valve disease, abdominal aortic aneurysm, and targeted cardiac therapies.
A key component of this effort is the use of the intrapericardial space as a distinct therapeutic compartment, enabling localized delivery of immunomodulatory, gene-targeting, and regenerative agents directly to the heart. This strategy reduces systemic exposure while enhancing mechanistic precision and translational relevance.
My research integrates targeted drug delivery, refined preclinical models, and immune-focused approaches to define and therapeutically modulate cardiovascular diseases.
Initiate a discussion